Researchers at the University of Virginia School of Medicine have identified a fundamental cause of chronic inflammation that accelerates aging, a discovery that could lead to longer, healthier lives and the possible avoidance of age-related illnesses such as lethal heart disease and debilitating brain problems.
Researchers discovered that the damaging inflammation is caused by incorrect calcium communication in the mitochondria of particular immune cells. Mitochondria provide energy in all cells and rely largely on calcium signaling.
Researchers at UVA Health, led by Bimal N. Desai, discovered that as humans age, mitochondria in immune cells known as macrophages lose their ability to take in and utilise calcium. This causes chronic inflammation, which is responsible for many of the disorders that plague us in our older years.
Researchers believe that boosting calcium uptake by mitochondrial macrophages could avoid inflammation and its disastrous consequences. Because macrophages are found in many organs of the body, including the brain, targeting them with proper medications may help halt the progression of age-related neurodegenerative disorders.
“I believe we have made a significant conceptual breakthrough in understanding the molecular underpinnings of age-related inflammation,” said Desai of UVA’s Department of Pharmacology and Carter Immunology Center.
According to Desai, the discovery offers possible therapy techniques for preventing inflammatory cascades, which are at the root of many cardiometabolic and neurological disorders. The study’s findings have been published in the journal Nature Aging.
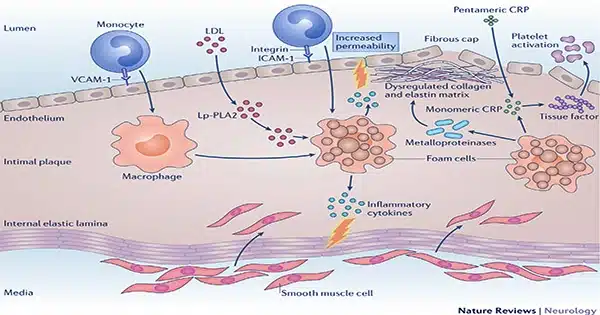
‘Inflammaging’ refers to the inflammation of aging. Macrophages are white blood cells that perform important roles in the immune system and in maintaining healthy health. They consume dead or dying cells, letting cellular detritus be removed, and they patrol for diseases and other foreign intruders. In this latter capacity, they serve as vital sentinels for the immune system, summoning assistance from other immune cells as needed.
Scientists have long known that macrophages lose effectiveness as they age, but it has been unclear why. Desai’s latest discovery proposes possible solutions.
Desai and his colleagues claim to have discovered a “keystone” mechanism responsible for age-related alterations in macrophages. Scientists believe that these modifications make macrophages prone to chronic, low-grade inflammation even under the best of circumstances. When challenged with an intruder or tissue damage, immune cells might become hyperactive. This causes “inflammaging”—chronic inflammation that accelerates aging.
Furthermore, the UVA Health researchers believe that the process they identified will apply not only to macrophages but to a wide range of additional immune cells created in the bone marrow. That means we may be able to encourage the appropriate functioning of those cells as well, potentially providing a significant boost to our immune systems as we age and grow more susceptible to disease.
Next steps: It will not be as straightforward as taking a calcium pill to treat inflammation. The issue isn’t so much a lack of calcium as it is the macrophages’ inability to utilize it appropriately. However, because Desai’s recent finding has identified the precise molecular machinery involved in this process, researchers should be able to find strategies to boost this machinery in aged cells.
Desai stated that the interdisciplinary scientific endeavor, which included computational biology, immunology, cell biology, and biophysics, would not have been possible without the perseverance of Phil Seegren, the graduate student who drove the study.
“Now, moving forward, we need an equally ambitious effort to figure out the wiring that controls this mitochondrial process in different types of macrophages and then manipulate that wiring in creative ways for biomedical impact,” Desai explained.