There were no atoms immediately after the Big Bang. Instead, what physicists call a hot soup of elementary particles filled the universe. It may not be long lasting, but this initial pillar helped shape what happened next – including us. Physicists have come to a significant conclusion in their search for an understanding of its nature; the “soup” flowed like water despite being unimaginably thick. Technically known as quark-gluon plasma, the first is believed to have filled the universe in microseconds after the universe was filled.
Costa Trachenko, a professor at Queen Mary University, is trying to reconstruct the nature of that plasma in a fraction of a second before cooling to allow atomic formation. He discovered its viscosity in SciPost physics. Quark-glue plasma is not just a thing of the past. It can exist today when the temperature is hot enough that the quarks that normally make protons and neutrons carry with them gluons that carry strong energy to mediate their interaction.
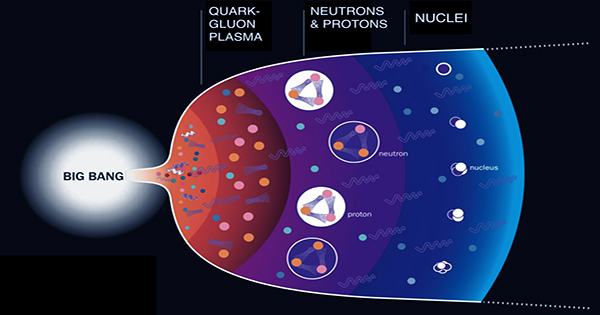
However, since it requires trillions of degrees of temperature (millions of times hotter than the center of the sun), it is not easy to study. We can, however, recreate it – albeit briefly – between particle collisions. Since the entire universe was still incredibly tightly packed, the original plasma-gluon soup was about 16 times (ten million billion) times less than water, Trachenko concluded, adding that he was interested at the time. Coincidentally, by a significant one, however, it was an order of magnitude 16 (more resistant to flow).
Since the flow of a liquid (somewhat mistakenly known as its kinematic viscosity) is determined by its dynamic viscosity ratio with its density, this means that plasma flowed at a rate equal to that of water. If taps were available at the time and anyone around could turn them on, hot and (relatively) cold running quark-gluon plasma could be on offer.
Putting the jokes aside, the investigation found that the particles in the plasma had gone away like water, increasing their ability to understand behavior. The exceptional properties of water, such as when it freezes, make it less dense, which is a constant source of wonder to physicists because a few other liquids divide them. That’s why it’s strange to find something so different with our most familiar substance.