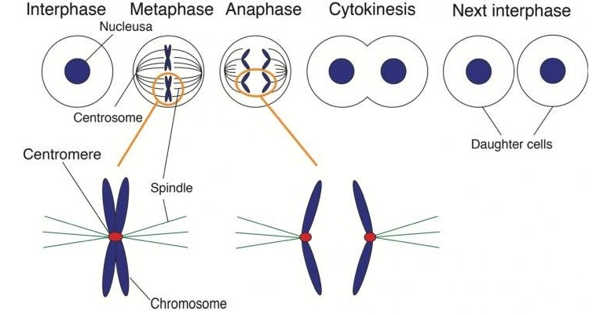
During cell division, chromosomes are duplicated and separated so that each of the two emerging daughter cells inherits one copy of each chromosome. Correct chromosome distribution necessitates high accuracy, and flaws in this process can result in aberrant chromosome distribution and facilitate cancer development. An international team led by scientists from the University of Geneva (UNIGE) has shed light on the mechanisms controlling this essential player in cell division by analyzing the structure of the protein responsible for chromosome separation. This research has been published in the journal Nature.
Before dividing, the cell duplicates its DNA, transforming it from a single chromosome with one arm to a double chromosome with two identical arms linked together by a ring-shaped protein complex called cohesin. The two arms are then separated by the action of separase, a molecular scissor that cuts a subunit of the cohesin complex to open up the ring.
Correct distribution of chromosomes requires high accuracy and defects in this process can cause aberrant distribution of chromosomes and facilitate cancer development.
The cell divides and gives birth to two identical daughter cells after the chromosomes are separated. Separase cleavage of cohesin is highly regulated and must occur only at a specific time during the cell cycle. To accomplish this, several inhibitory proteins independently block separase activity until the chromosomes must be separated. However, the molecular mechanisms by which inhibitors control separase activity have remained a mystery until now.
To ensure high fidelity, chromosome segregation is a mechanical process that relies on the structural integrity of the microtubule spindle apparatus and a checkpoint signaling pathway. Another complicated process occurs when the cell ensures that exactly one set of duplicated chromosomes is transferred to each of the two cells produced during cell division.
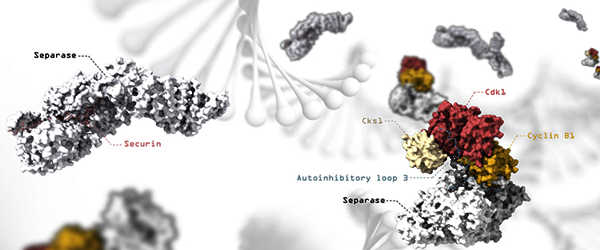
High-resolution electron microscopy used to reveal regulatory mechanisms
The scientists used cryogenic electron microscopy in this study led by Andreas Boland, professor in the Department of Molecular Biology at the UNIGE Faculty of Science (cryoEM). “This technique allows us to observe biological samples at very high resolution while keeping them in their natural state,” explains Jun Yu, first author of this study and a researcher in the Department of Molecular Biology.
They were able to determine several structures of human separase in complex with one of its inhibitors using this method, revealing new regulatory mechanisms for the enzyme. “It turns out that these inhibitors occupy sites that also recognize the cohesin substrate, preventing the molecular scissors from cleaving,” Andreas Boland explains.
Inhibiting a protein by changing its conformation
While one of the inhibitors, securin, binds directly to the molecular scissors to block its active site, another, the CCC complex, works through a more complex mechanism. The CCC complex causes a conformational change in separase by binding to its periphery. As a result, separase loops, which are normally flexible and disordered, are reorganized into a fixed position, causing the enzyme to self-inhibit.
“Our work significantly contributes to our understanding of the mechanisms that regulate separase activation and may aid in the development of novel anti-cancer therapies,” Andreas Boland concludes.
Every living organism relies on chromosome segregation to maintain genome stability during mitotic and meiotic divisions. Chromosomes ensure correct segregation by attaching to the mitotic spindle via the kinetochore, a large proteinaceous complex that forms at centromeric chromatin during the onset of mitosis.