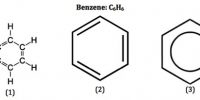
When a coal decomposes (‘carbonises’) there are three classes of product: solid, liquid and gaseous. The solid we refer to as coke or as char. The gases include hydrogen and carbon monoxide. The liquid product is known as coal tar. Carbonization of coal on the industrial scale began in the late eighteenth century. At that time charcoal was being used to make iron from iron ore, in response to the huge demand for iron brought about by industrialization. Much of the south of England had until that time been heavily forested and very conspicuous gaps were created through the removal of the trees for charcoal production. Over time coke replaced charcoal as a metallurgical reductant.
Steel manufacturers naturally tended to establish themselves dose to coalfields. The conurbation of Sheffield, Rotherham and Barnsley is probably the best example of this in Britain. The best example in the US is Pittsburgh.
When coke is made, gas and tar are always by-products. The gas became used in lighting, there were gas lamps in London by the 1820s. Later, gas was reticulated to homes; by the end of the nineteenth century many homes in the UK had gas ‘laid on; still largely for illumination as an alternative to candles. The star product of coal carbonization became feedstock for the very earliest organic chemistry industry. Germany was the leading nation in this respect. Coal tar as a source of chemicals continued until the 1920s at which stage it was replaced by cracked hydrocarbon products. The author has made elsewhere the comment that the development of cracking technologies and cessation of reliance on coal tar revolutionized the organic chemistry industry.
From then on the US, especially in the Gulf Coast states, became the leader though not to the exclusion of Germany. Many companies including IG Farben, BASF and Hoechst were active in this respect in both countries.