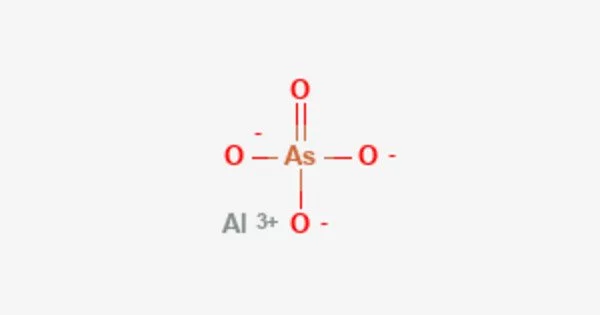
Aluminium arsenate is a chemical compound. It is an inorganic compound with the formula AlAsO4. It is most commonly found as an octahydrate. It is a colourless solid that is produced by the reaction between sodium arsenate and a soluble aluminium salt. Aluminium arsenate occurs naturally as the mineral mansfieldite. Anhydrous form is known as an extremely rare, fumarolic mineral alarsite. A synthetic hydrate of aluminium arsenate is produced by hydrothermal method.
It is a common impurity in aluminium compounds and can be toxic if ingested in large quantities. It is not a commonly used chemical in industry and research, and there is limited information available about its properties and uses.
Properties
It is a white, powdery substance that is highly insoluble in water. It is not commonly used in industry or in consumer products. However, it is used in the production of pigments for ceramics, glass, and paint. It is also used as a fungicide in agriculture. It is toxic if inhaled or ingested and should be handled with care.
- Chemical formula: AlAsO4
- Molar mass: 165.899 g·mol−1
- Appearance: colourless crystals
- Density: 3.25 g/cm3
- Melting point: 1,000 °C (1,830 °F; 1,270 K)
- Solubility in water: insoluble
- Crystal structure: hexagonal
Modification of aluminium orthoarsenate was carried out by heating different samples to different temperatures. Both amorphous and crystalline forms were obtained. The solubility product was determined to be 10−18.06 for aluminium arsenate hydrate of formula AlAsO4·3.5H2O. Like gallium arsenate and boron arsenate, it adopts the α-quartz-type structure. The high-pressure form has a rutile-type structure in which aluminium and arsenic are six-coordinate.
Application
It is a white or colourless powder that is insoluble in water. It is a rare mineral and is not known to have any commercial or industrial uses. Some studies have shown that it has antifungal properties, but its toxicity is not well understood. It is a white powder that is insoluble in water. It is used as a pesticide and as a starting material for the production of other aluminum compounds. It is toxic if inhaled or ingested and should be handled with care.
It is typically found in the form of a white powder and is used in a variety of industrial applications, such as in the production of pesticides and wood preservatives. However, aluminum arsenate is highly toxic and can be harmful if ingested or inhaled, so it must be handled with care.