Bromine pentafluoride, BrF5, is a bromine fluoride and an interhalogen compound. It has high fluorinating power. It has the appearance of a colorless, fuming liquid with a strong odor. It is used in the production of other chemicals as well as rockets. It is extremely toxic when inhaled. It corrodes metals and tissue. It will hasten the combustion of combustible material. If the containers catch fire, they may explode violently and rocket.
BrF5 is used in the analysis of oxygen isotopes. In the presence of BrF5, laser ablation of solid silicates releases O2 for subsequent analysis. It has also been used as an oxidizer in liquid rocket propellants and as a fluorinating agent in uranium processing.
Properties
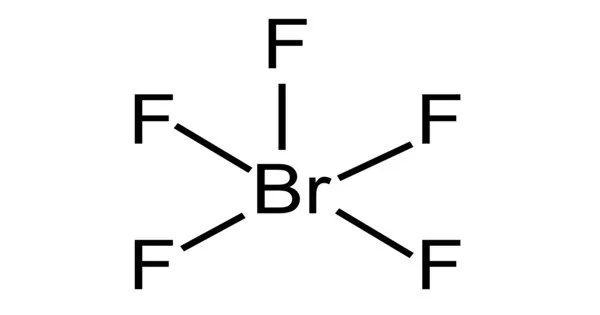
Bromine pentafluoride is a colorless, corrosive liquid, used as an oxidizer in liquid rocket propellants. At temperatures above boiling point this chemical is a colorless gas. It is an interhalogen compound and a fluoride of bromine. The molecule has a central bromine atom that is surrounded by five fluorides and a lone pair of electrons.
- Chemical formula: BrF5
- Molar mass: 174.894 g.mol−1
- Appearance: Pale yellow liquid
- Density: 2.466 g/cm3
- Melting point: −61.30 °C (−78.34 °F; 211.85 K)
- Boiling point: 40.25 °C (104.45 °F; 313.40 K)
- Solubility in water: Reacts with water
- Molecular shape: Square pyramidal
Preparation
Bromine pentafluoride is prepared by fluorination of bromine at 200°C. The reaction is carried out in an iron or copper vessel. The halogens are diluted in nitrogen. BrF5 was first prepared in 1931 by the direct reaction of bromine and fluorine. This reaction is suitable for the preparation of large quantities, and is carried out at temperatures over 150 °C (302°F) with an excess of fluorine:
Br2 + 5 F2 → 2 BrF5
For the preparation of smaller amounts, potassium bromide is used:
KBr + 3 F2 → KF + BrF5
This route yields BrF5 almost completely free of trifluorides and other impurities.
Reactions
BrF5 reacts with water to form bromic acid and hydrofluoric acid:
BrF5 + 3 H2O → HBrO3 + 5 HF
It is an extremely effective fluorinating agent, being able to convert most metals to their highest fluorides even at room temperature. With uranium and uranium compounds, it can be used to produce uranium hexafluoride:
5 U + 6 BrF5 → 5 UF6 + 3 Br
Hazards
BrF5 has a strong corrosive and toxic reaction with water. Its vapors are also highly irritating to all parts of the human body, particularly the skin, eyes, and mucous membranes. When exposed to moist air, it, like many other interhalogen compounds, will emit “smoke” containing acidic vapors as a result of its reaction with the water in the air. Most experimental animals die after being exposed to 100 ppm or higher for more than one minute. Chronic exposure can lead to kidney and liver damage.
Furthermore, BrF5 is a strong oxidizing agent that can spontaneously ignite or explode when it comes into contact with flammable substances like organic materials and metal dust.