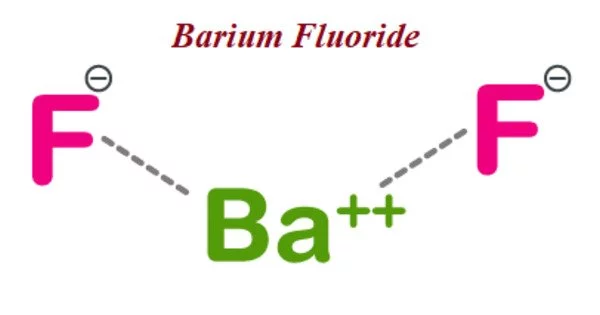
The formula for barium fluoride is BaF2, is an inorganic compound. It is a colorless solid that occurs naturally in the form of the rare mineral frankdicksonite. It takes on the fluorite structure under normal conditions and the PbCl2 structure under high pressure. It is resistant to and insoluble in water, just like CaF2. It can be found in nature as a mineral called frankdicksonite.
Moisture corrodes BaF2 above around 500 °C, but in dry environments it can be used up to 800 °C. Long-term moisture exposure reduces transmission in the vacuum UV range. It is less resistant to water than calcium fluoride, but it is the most resistant of all the optical fluorides to high-energy radiation, though its far ultraviolet transmittance is lower than that of the other fluorides. It is quite hard, very sensitive to thermal shock and fractures quite easily.
Properties
The melting point of the barium fluoride is 1,368 °C, whereas, it has a boiling point of 2,260 °C. It carries a density of 4.89 g/cm³. Moreover, the molar mass of the barium fluoride is 175.34 g/mol. It is slightly soluble in the water (H2O).
- Chemical formula: BaF2
- Molar mass: 175.324 g/mol
- Appearance: white cubic crystals
- Density: 4.893 g/cm3
- Melting point: 1,368 °C (2,494 °F; 1,641 K)
- Boiling point: 2,260 °C (4,100 °F; 2,530 K)
- Solubility in water: 1.58 g/L (10 °C); 1.61 g/L (25 °C)
- Solubility product (Ksp): 1.84 × 10−7
- Solubility: soluble in methanol, ethanol
- Thermal conductivity: 10.9 W/(m·K)

Optical properties
From 150-200 nm to 11-11.5 µm, barium fluoride is transparent from the ultraviolet to the infrared. It is used in windows for infrared spectroscopy, particularly in the field of fuel oil analysis. Its transmittance is relatively low at 200 nm (0.60), but at 500 nm it rises to 0.96-0.97 and remains there until 9 µm, when it begins to fall (0.85 for 10 m and 0.42 for 12 µm). From 700 nm to 5 µm, the refractive index is about 1.46.
Barium fluoride is also a widely used, very fast (one of the fastest) scintillator for detecting X-rays, gamma rays, and other high energy particles. Its detection of 511 keV gamma photons in positron emission tomography is one of its applications. It reacts to alpha and beta particles as well, but unlike most scintillators, it does not emit ultraviolet light. It can also be used to detect high-energy (10-150 MeV) neutrons by using pulse shape discrimination techniques to distinguish them from concurrently occurring gamma photons.
Gas phase structure
The BaF2 molecule is non-linear in the vapor phase, with an F-Ba-F angle of about 108°. Its nonlinearity contradicts VSEPR theory. According to ab initio calculations, contributions from d orbitals in the shell below the valence shell are to blame. Another theory is that polarisation of the electron core of the barium atom results in a roughly tetrahedral charge distribution that interacts with the Ba-F bonds.
Uses
Barium fluoride is used as a preopacifying agent, as well as in the manufacture of enamel and glazing frits. It is also used in the manufacture of welding agents (an additive to some fluxes, a component of coatings for welding rods and in welding powders). It is also used as a molten bath for refining aluminum in metallurgy.
- Used to make optical components such as lens,
- Used in the manufacture of carbon brushes for DC motor,
- Used in the manufacture of glass.