Abnormal Molecular Masses from Colligative Properties from Association in solution
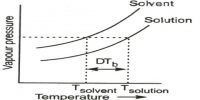
The laws of the colligative properties are applicable only for very dilute solution, and when there are no association and dissociation of the solutes in solution. Sometimes abnormal molecular masses of the solutes were observed when calculated from the laws of colligative properties. Let us consider association and dissociation of solutes in solutions.
Association in solution: Compounds like ethanoic acid, phenol, benzoic and etc., in benzene solutions give values of osmotic pressure, freezing point depression, and boiling point elevation much lower than those expected from the respective equations. Consequently, the molecular masses derived from these values were higher than those indicated by the molecular formula. This was explained by assuming association of solute molecules to form aggregates having large molecular mass. Acetic acid and benzoic acid are known to form dimmers at higher concentrations and the existence of an equilibrium of the type 2A ↔ (A)2 at appreciable concentration has been predicted.