Determination of Critical Constants
Long before Andrews performed his experiments with carbon dioxide, Caignard de La Tour (1822) observed that when ether was heated in a sealed tube, the surface of separation between liquid and its vapor disappeared at a particular temperature; but on cooling the surface reappeared at about the same temperature. The significance of these observations was not realized until Andrews correlated this with his work on carbon dioxide. He observed that the temperature at which the surface of separation between liquid and its vapor disappears is the same at which he observed the Critical phenomenon.
These observations have been utilized for the determination of the critical constants. Critical pressure and temperature may be determined fairly easily by using an apparatus as shown in the figure; similar to Caignard de La Tour’s apparatus.
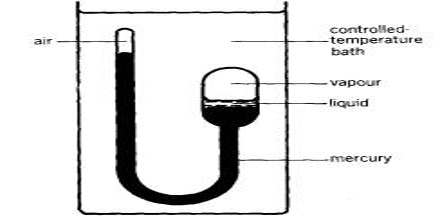
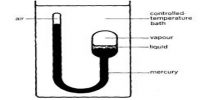
It consists of a thick-walled glass U-tube one limb of which is longer than the other. The liquid is contained in the bulb and the rest of the apparatus is filled with mercury and the upper and of the bulb is preserved leaving a little air in it. The shorter end containing the liquid in contact with its saturated vapor is sealed and is surrounded by a heating jacket. The other limb of the U-tube, which serves as a manometer, has aired at its upper-end A, the rest of the tube being filled with mercury.
The temperature of the bulb is raised gradually. The end B is gradually heated by passing hot air or vapor through the jacket and the temperature at which the meniscus separating liquid and vapor just disappears and at which it reappears on cooling is noted. On cooling this mixture, a mist first forms and the liquid appear quickly. The average of the two is the critical temperature. The volume of the air in the limb A is also noted at this temperature and the critical pressure is calculated from this. The critical temperature, (Tc), is characteristic of every gas and might be defined as: “The temperature beneath which the constant enlarge of pressure on gas ultimately brings about liquefaction and above which no liquefaction can take place no issue what so ever pressure be applied”.
When the liquid and its vapor are gradually heated the density of the liquid decreases, but the density of the vapor increases, because of compression until at the critical temperature and pressure the densities of both liquid and vapor, become equal, and the liquid and vapor become indistinguishable from each other. The mean of the temperature of the evaporation and return of the liquid in the bulb is critical temperature. The pressure read on the manometer is “critical pressure”. The pressure required to liquefy the gas at critical temperature is called critical pressure and the volume occupied by 1 mole of gas under these conditions is called the critical volume. This is the reason why at the critical point the surface of separation vanishes. The critical constants of liquid explosives cannot be calculated in a straight line because of the obdurate nature of the substances concerned.