Ideal and Non – ideal Solutions
One property of completely miscible liquid pairs is the energy change involved when two liquids are mixed. We know that when one liquid dissolves in another we can imagine that the molecules of the solvent are caused to move apart so as to make room for the molecules of the solute. Since there are attractive forces between the molecules of the solute and those of the solvent, energy has to be added in order to separate from one another the molecules of the solvent as well as of the solute. When the molecules of the solvent and solute come together to form the solution, energy is released because of the attractions between the molecules of the solvent and the solute.
Let consider three different scenarios;
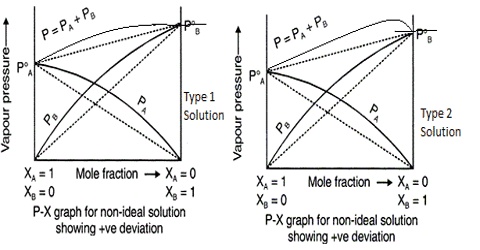
(a) The attractive forces between the molecules of the solute, between the molecules of the solvent and between the molecules of solvent and of the solute are similar, as in the solution of hexane and octane. The energy absorbed in the separation of the molecules of the solvent and the molecules at the solute becomes equal to the energy released when the solvent and solute molecules mix together to form the solution. No heat is absorbed or released on mixing the two liquids. Such a solution is called an ideal solution. Such solutions obey Raoult’s law. A solution is said to be ideal when it obeys Raoult’s law over all compositions and at all temperatures.
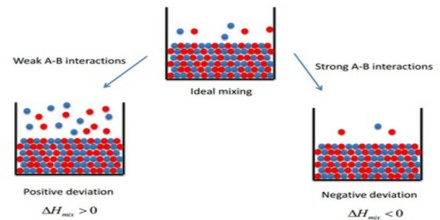
(b) If the molecules of the solvent and the solute attract each other more strongly than molecules of their own kind, more energy can be released when the solute and solvent molecules an brought together than was required to separate them. Under these circumstances the overall solution process can result in the evolution of heat and the mixing will be exothermic. When acetone and water are mixed heat is evolved and the container in which the mixing is done becomes warm.
(c) When the salute – Solvent molecular interaction is weaker than those between pure solvent molecules and pure solute molecules the formation of a solution requires addition of energy. This is became more energy is required to separate the molecules in the solute and solvent that is evolved when these molecules are mixed. In such cases the solution becomes cool as it is formed, indicating that the process is endothermic. Solution of ethanol and hexane is an example. The non- polar hexane molecules come between ethanol molecules effectively destroying the hydrogen bond between ethanol molecules. This absorbs energy and the solution is cool.
Liquid – liquid solution of the types described in (b) and (c) are non-ideal as these do not obey Raoult’s law.