Molecular Theory of Surface Tension:
Consider two molecules P and Q as shown in Figure. Taking them as centers and molecular range as radius, a sphere of influence is drawn around them.
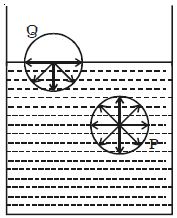
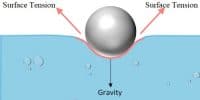
The molecule P is attracted in all directions equally by neighboring molecules. Therefore net force acting on P is zero. The molecule Q is on the free surface of the liquid. It experiences a net downward force because the number of molecules in the lower half of the sphere is more and the upper half is completely outside the surface of the liquid. Therefore all the molecules lying on the surface of a liquid experience only a net downward force.
If a molecule from the interior is to be brought to the surface of the liquid. work must be done against this downward force. This work done on the molecule is stored as potential energy. For equilibrium, a system must possess minimum potential energy. So, the free surface will have minimum potential energy. The free surface of a liquid tends to assume minimum surface area by contracting and remains in a state of tension like a stretched elastic membrane.