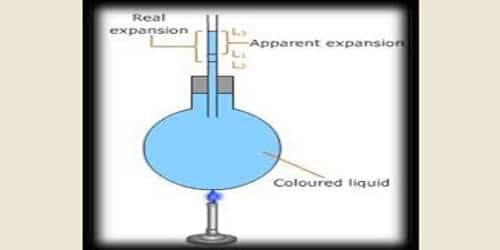
If the liquid is heated directly without using any container then the expansion that you observe is termed as a real expansion of the liquid. The expansion of the liquid apparently observed without considering the expansion of the container is called the apparent expansion of the liquid. If we consider the expansion of the container also and measure the total expansion in the volume of the liquid, then the expansion is termed as the absolute expansion of the liquid.
Explanation
A glass bulb with a long graduated stem is filled with liquid up to the mark A. Now keeping an eye on the liquid column the bulb is heated and it is observed that the upper level of the liquid comes down from position A to position B. After that, it moves up from the graduated line B crossing the mark A and reaches to mark C.
The reason is that when the heat is applied the volume of the bulb increases at first. Due to this cause liquid comes down to B from A. Later on as soon as the liquid gets heated its volume starts to increase and reaches from B to C. It happens so as the expansion of liquid is more than the expansion of solid.
Apparently it appears to us that the liquid was at mark A and finally reaches to mark C. Therefore, CA is the apparent expansion. CB is the real expansion and AB indicates the expansion of the container.
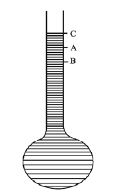
From the figure it is observed that,
CB = CA+AB
Real expansion = Apparent expansion + expansion of the container
So, Vr = Va + Vg
Unlike solids, liquids have no fixed length or surface area but always take up the shape of the containing vessel. When a liquid is heated in a container, heat flows through the container to the liquid; which means that the container expands first, due to which the level of the liquid falls. When the liquid gets heated, it expands more, beyond its unique level. We cannot monitor the intermediate state. We can only observe the initial and final levels. This observed expansion of the liquid is known as the apparent expansion of the liquid.
The real expansion of liquid = Apparent expansion of liquid + Volume expansion of the container.
Therefore, in the case of liquids, we are concerned only with volume changes when they are heated. The real (or absolute) expansivity of a liquid is the fraction of its volume by which it expands per kelvin rise in temperature.