For Earth-bound explorers, water scarcity – especially potable water – was often the biggest obstacle. Long-distance travelers will not only need to drink water but it will also be used to create oxygen. However, the techniques we use to split water on Earth run into problems outside the strong gravitational field. Luckily, it looks like we have the answer now. Earthly free oxygen is a product of plants and photosynthesizing bacteria. Future space travel can be carried with us to ensure that astronauts can breathe, but the required weight and space can prove too much.
The International Space Station receives its oxygen from the electrolyzing water, through which electricity is powered so that oxygen and the other hydrogen are present, but there is room for improvement in the process. Long space missions will not be able to stock up on water and their energy sources will weaken as they travel further from the sun, so Caltech’s Dr. Katharina Brinkert is looking for a lighter and more efficient alternative to turn it into oxygen.
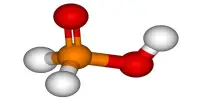
On Earth, when we use electricity to split water to make hydrogen for fuel, the process works because the gases bubble on the surface on which they can be trapped. In microgravity, the gases do not increase uniformly, resulting in increased resistance to gas bubble froth layers and gradual gas production. One option is to create artificial gravity, spinning the chamber where the water is splitting like a centrifuge. In fact, future journeys to outer planets can do this with the entire spacecraft for the comfort of the gathering. However, Brinkert wants to make sure that options are available.
In nature communication, he announced a small scale solution. Brinkert and colleagues used the Bremen drop tower to mimic the gravity of zero, although their equipment fell for free for only 9.3 seconds. The water-dividing cells were camera-held together to record the development of hydrogen gas bubbles in one-electrode and the other in oxygen.
Brinkart trails two electrolysis systems, both using indium phosphide, and rhodium particles. These brought very similar results under the Earth’s gravity, but a million times less performance under gravity. The other system performed just as well with less gravity.
Unfortunately, Elon Musk (due to his public disdain for nanotechnology) had to self-assemble nanocrystalline rhodium particles in hexagonal cell patterns in order to use the appropriate version of his planned orbit on Brinkert’s Mars. nanostructuring hydrogen bubbles prevent the current from becoming large enough to induce erosion, which further interferes with the separation of water. In addition to remote space missions, this work could lead to better design for the separation of water on Earth, where the goal is to store hydrogen in clean energy.