Aluminium diboride (AlB2) is a chemical compound formed from the elements aluminium and boron. It is one of two aluminium-boron compounds, the other being AlB12, that are known collectively as aluminium boride. It is a member of the metal boride family and is known for its intriguing physical and chemical properties. It is a hard material with a hardness comparable to tungsten carbide.
Properties
Aluminum diboride has a high melting point, making it useful in applications where materials are exposed to high temperatures. It is an electrical conductor, making it suitable for certain applications in electronics. It is often used as a ceramic material. It is sometimes combined with other materials to form ceramic composites with enhanced properties.
- Chemical formula: AlB2
- Molar mass: 48.604 g/mol
- Appearance: Copper-red solid
- Density: 3.19 g/cm3
- Melting point: >920 °C (decomposes)
- Solubility in water: insoluble
- Crystal structure: Hexagonal, hP3
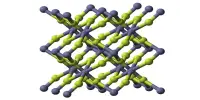
Crystal Structure
Aluminum diboride has a hexagonal crystal structure. The hexagonal lattice is made up of alternating aluminum and boron atoms. The B atoms form graphite-like sheets with Al atoms between them, which are very similar to the structure of magnesium diboride. Metallic conductivity is observed in single crystals of AlB2 along an axis parallel to the basal hexagonal plane.
The crystal structure of AlB2 is frequently used as a prototype structure to describe intermetallic compounds. The AlB2 structural family includes a wide range of structure types.
Applications
- Abrasive Applications: Due to its hardness, it is used in abrasive applications.
- High-Temperature Applications: Its high melting point makes it suitable for applications involving high temperatures.
- Ceramic Composites: It is used as a component in ceramic composites, where its properties contribute to the overall performance of the material.
Safety
Aluminium boride is considered a hazardous substance as it reacts with acids and hydrogen gas to produce toxic gases. For example, it reacts with hydrochloric acid to release borane and aluminium chloride.