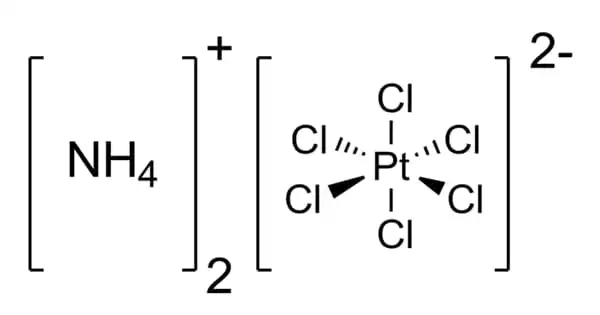
Ammonium hexachloroplatinate, also known as ammonium chloroplatinate, is the inorganic compound with the formula (NH4)2[PtCl6]. It is an ammonium salt comprising separate ammonium cations and octahedral [PtCl6](2-) anions. It is a rare example of a soluble platinum(IV) salt that is not hygroscopic. It contains a hexachloroplatinate(2-). It forms intensely yellow solutions in water. In the presence of 1M NH4Cl, its solubility is only 0.0028 g/100 mL.
Properties
It is a rare example of a soluble platinum(IV) salt that is not hygroscopic. It forms intensely yellow solutions in water. In the presence of 1M NH4Cl, its solubility is only 0.0028g/100 mL.
- Molecular Weight: 443.87
- Appearance: Yellow to orange powder or crystals
- Melting Point: 380 °C (decomposes)
- Boiling Point: N/A
- Density: 3.07 g/cm3
- Solubility in H2O: N/A

Preparation and structure
The compound consists of separate tetrahedral ammonium cations and octahedral [PtCl6]2- anions. It is usually generated as a fine yellow precipitate by treating a solution of hexachloroplatinic acid with a solution of an ammonium salt. The complex is so poorly soluble that this step is employed in the isolation of platinum from ores and recycled residues.
As analyzed by X-ray crystallography, the salt crystallizes in a cubic motif reminiscent of the fluorite structure. The [PtCl6]2- centers are octahedral. The NH4+ centers are hydrogen bonded to the chloride ligands.
Uses and reactions
Ammonium hexachloroplatinate is used in platinum plating. Heating (NH4)2[PtCl6] under a stream of hydrogen at 200 °C produces platinum sponge. Treating this with chlorine gives H2[PtCl6].