The inorganic compound with the formula H2Te is hydrogen telluride. It is a colourless gas that is a hydrogen chalcogenide and the simplest tellurium hydride. It is a binary hydride and a member of the chalcogen hydrides group, which includes periodic table compounds of hydrogen with elements from the oxygen family (Group 16).
Although unstable in ambient air, the gas can exist at extremely low concentrations long enough to be detected by the odour of rotting garlic at extremely low concentrations or the revolting odour of rotting leeks at slightly higher concentrations. It is highly unstable and, when exposed to light or heat, decomposes into its constituent elements hydrogen (H2) and tellurium (Te). Due to its unstable and toxic nature, it is not encountered in significant quantities in nature and is typically synthesized in laboratories for specific purposes.
Properties
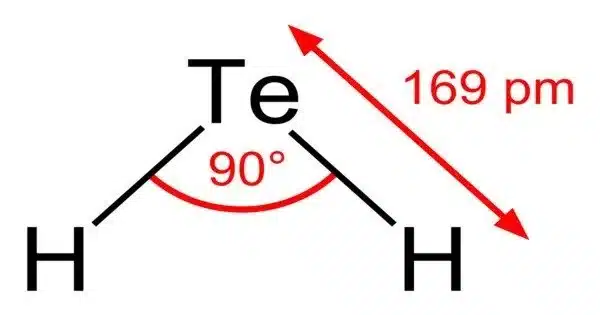
Most compounds with Te–H bonds (tellurols) are unstable with respect to loss of H2. H2Te is chemically and structurally similar to hydrogen selenide, both are acidic. The H–Te–H angle is about 90°. Volatile tellurium compounds often have unpleasant odours, reminiscent of decayed leeks or garlic.
- Chemical formula: H2Te
- Molar mass: 129.6158 g mol−1
- Appearance: colourless gas
- Odor: Pungent, resembles rotting garlic or leeks
- Density: 3.310 g/L, gas 2.57 g/cm3 (−20 °C, liquid)
- Melting point: −49 °C (−56 °F; 224 K)
- Boiling point: −2.2 °C (28.0 °F; 270.9 K) (unstable above −2 °C)
- Solubility in water: 0.70 g/100 mL
- Acidity (pKa): 2.6
- Conjugate acid: Telluronium
- Conjugate base: Telluride
- Molecular shape: bent
Reactions
H2Te is an endothermic compound, degrading to the elements at room temperature:
H2Te → H2 + Te
Light accelerates the decomposition. It is unstable in air, being oxidized to water and elemental tellurium:[6]
2 H2Te + O2 → 2 H2O + 2 Te
It is almost as acidic as phosphoric acid (Ka = 8.1×10−3), having a Ka value of about 2.3×10−3. It reacts with many metals to form tellurides.
Application
Because of its instability and toxicity, hydrogen telluride has few practical applications. However, it can be used in research and certain chemical processes where tellurium’s properties are desired. Nonetheless, handling this substance necessitates extreme caution and appropriate safety precautions. When working with hydrogen telluride, it is critical to use proper laboratory equipment and protective equipment.
The gas has a foul odour similar to rotten eggs, which is characteristic of many tellurium-containing compounds. Because of its toxicity, inhaling hydrogen telluride should be avoided because it can cause serious health problems.