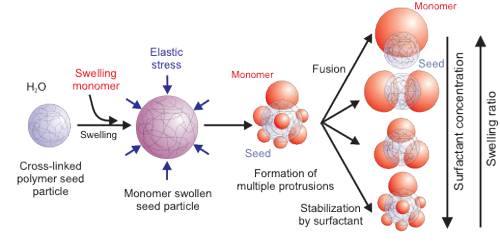
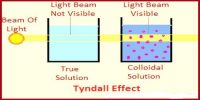
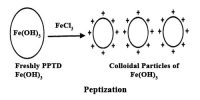
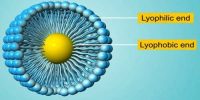
Shape of colloidal particles
The colloids do not have any definite and uniform shape. Colloidal refers to items of a small size that are floating in a medium of one of three substances: a solid, a liquid, or a gas. A large number of lyophobic colloids are spherical or nearly spherical as can be seen in a high power microscope. Colloidal particles can be suspended in a substance just like their own makeup or a different substance with the exception of gases.
However, non-spherical particles like rods or discs are also sometimes observed but their occurrence is rare. Most lyophobic colloids are not perfect spheres, ellipsoids, rods or discs. They have intermediate shapes. For sub-microscopic particles, the shape can be estimated to some extent from the twinkle of the particles in an ultramicroscope. Careful observation is necessary. The shape of some colloids can be fairly well ascertained from their appearance in stirred and unstirred conditions. Thus a number of sols, when stirred, show characteristic ‘silky’ appearance. This is only possible if the particles are rod-like or disc-shaped. Vanadium pentoxide sol is an example. Association colloids have definite shapes for a particular system but disintegration colloids may vary widely in shape. They are often of irregular shape.
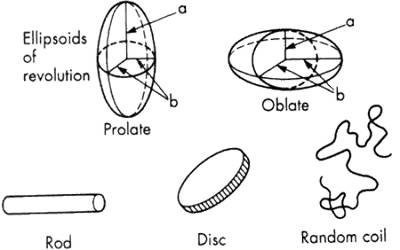
However, for many theoretical treatments, it becomes necessary to assume a definite geometrical shape, usually a sphere, for the sake of simplicity. Molecular colloids also have no definite shape in solution. Some are almost spherical while some are rod-like. Many linear high polymers have a rod-like shape. Several proteins also are of similar shape. The shape of high polymers may vary with the molecular mass. Thus a stiff rod-like molecule may become deformed due to increase in flexibility on increase of molecular mass and may behave like a random coil. One of the principal objects, of the physical chemists working in the high polymer field is to ascertain the shape of the macromolecules. The practical application of colloids in many fields depends on their shape, and whether the colloid is lyophobic or lyophilic. Thus the final finish of a surface coated with paint or varnish will depend on the shape of the particles. Only linear high polymers are suitable for textile purposes whereas the plastic type of materials is best made of non-linear cross-linked polymers.