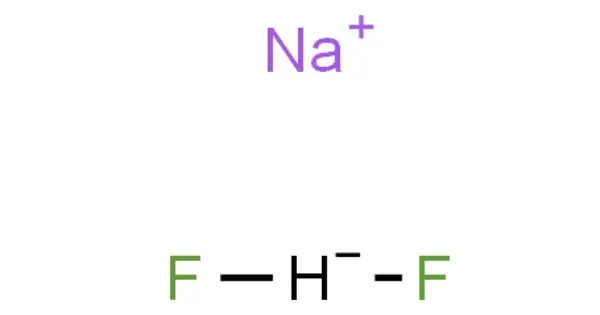
Sodium bifluoride is an inorganic chemical with the formula Na[HF2. It is a salt composed of sodium cation (Na+) and bifluoride anion ([HF2]−). It is a chemical compound made up of sodium, hydrogen, and fluoride. It is a white, water-soluble substance that decomposes when heated. Sodium bifluoride is nonflammable, hygroscopic, and has a strong odor. Sodium bifluoride has numerous industrial applications. It is a white, crystalline substance that is extremely soluble in water.
Properties
- Chemical formula: Na[HF2]
- Molar mass: 61.995 g·mol−1
- Appearance: white solid
- Density: 2.08 g/cm3
- Melting point: 160 °C (320 °F; 433 K) (decomposes)
Reactions
Sodium bifluoride dissociates to hydrofluoric acid and sodium fluoride:
Na[HF2] ⇌ HF + NaF
This reaction is reversed to remove HF from elemental fluorine (F2) produced by electrolysis. This equilibrium is achieved when the salt is dissolved and the solid is heated. Like other bifluorides, it interacts with acids to produce HF. The reaction with bisulfate, for example, produces sodium sulfate and hydrogen fluoride.
Strong bases deprotonate bifluoride. For example, calcium hydroxide produces calcium fluoride.
Production
Sodium bifluoride is produced by neutralizing waste hydrogen fluoride, which results from the production of superphosphate fertilizers. Typical bases are sodium carbonate and sodium hydroxide. The process occurs in two steps, illustrated with the hydroxide:
HF + NaOH → NaF + H2O
HF + NaF → Na[HF2]
Sodium bifluoride reacts with water or moist skin to form hydrofluoric acid. It also emits hydrofluoric acid and hydrogen gas when heated to a gaseous state. When the chemical comes into touch with strong acids or bases, metal, water, or glass, it decomposes. Sodium bifluoride also reacts violently with chromium chloride, nitric acid, red phosphorus, sodium peroxide, diethyl sulfoxide, and diethylzinc.
Applications
Sodium bifluoride serves primarily as a precursor to sodium fluoride, which is manufactured in millions of tons each year. It is utilized in several industrial operations, including as metal surface treatment and cleaning. It is used in the synthesis of some fluorine compounds. It can be used to recover beryllium from beryllium ore.
Precautions
Sodium bifluoride is corrosive and irritating to the skin, causing blistering and irritation. It’s incredibly harmful to consume. If the compound enters the eyes, it can cause blindness and corneal damage. Ingesting sodium bifluoride dust can induce burning, coughing, and sneezing by irritating the gastrointestinal and respiratory systems. Exposure to the chemical in the eyes might produce redness, irritation, and watering. In severe situations, exposure to sodium bifluoride might lead to death. The symptoms of sodium bifluoride poisoning might take anywhere from 0 to 24 hours to appear.