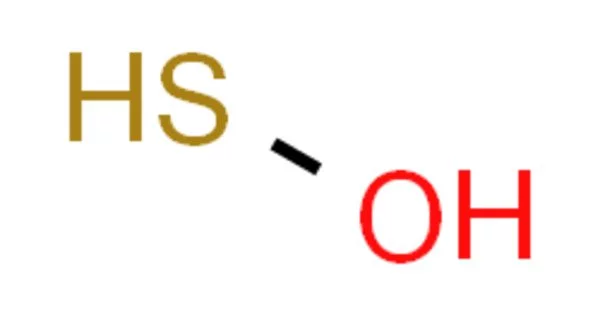Hydrogen thioperoxide, also known as oxadisulfane, is a chemical with the formula H-S-O-H. It is the simple sulfur-substituted analog of the common hydrogen peroxide (H-O-O-H) chemical, as well as the simplest hydrogen chalcogenide containing more than one type of chalcogen.
The chemical has been described as the “missing link” between hydrogen peroxide and hydrogen disulfide (H-S-S-H), though it is significantly less stable than either of the other two. It is the inorganic parent structure of the sulfenic acid class of organic compounds (R-S-O-H) as well as the oxadisulfide linkage (R1-S-O-R2), where “R” can be any organic structure. Sulfur is present in oxidation state 0.
Properties
Hydrogen thioperoxide molecules have a gauche conformation. They are unsymmetrical but have a low barrier to convert from left-hand to right-hand forms so that the molecule can tunnel between the forms.
The measurements of the bond-lengths in hydrogen thioperoxide are H–S 1.3420 Å, S–O 1.6616 Å, O–H 0.9606 Å. The bond angles are H–S–O 98.57°, S–O–H 107.19°. The H–S and O–H bonds are twisted at 90.41°.
- Chemical formula: H2OS
- Molar mass: 50.08 g·mol−1
- Density: 1.249
Formation
Hydrogen thioperoxide has been synthesized in laboratories through photolysis of an ozone-hydrogen sulfide mixture frozen in argon at 8 K and pyrolysis of di-tert-butyl sulfoxide. Another method is to conduct an electric discharge through water and sulfur.
In the interstellar medium, hydrogen thioperoxide is thought to be formed by the reaction of sulfur monoxide with the trihydrogen cation, dihydrogen, and an electron. Another route is for sulfur monoxide to react with atomic hydrogen to form HOS and HSO, which can then add another hydrogen atom. However, this mechanism is likely to require the removal of excess energy by a dust grain.
Reactions
Two molecules of hydrogen thioperoxide can undergo cyclocondensation to form sulfinothioic acid HS(=O)SH and water.
Hydrosulfide HS− can react with HSOH to yield disulfane HSSH.