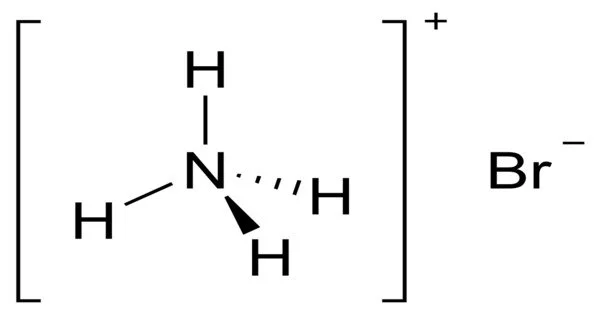
The ammonium salt of hydrobromic acid, NH4Br, is ammonium bromide. The chemical crystallizes in colorless prisms with a saline taste; when heated, it sublimes and is easily soluble in water. When exposed to air, it gradually turns yellow due to the oxidation of traces of bromide (Br–) to bromine (Br2). It is made up of atoms of various elements combined in a specific ratio.
Ammonium bromide is used in photography in films, plates, and papers, as well as in wood fireproofing, lithography and process engraving, corrosion inhibitors, and pharmaceutical preparations.
Properties
Ammonium Bromide is odorless white crystals or granules which become yellow when exposed with air. It is the ammonium salt of hydrobromic acid. It is soluble in water. On exposure to air, it becomes to yellow color because of the oxidation of traces of bromide to bromine.
- Chemical formula: NH4Br
- Molar mass: 97.94 g/mol
- Appearance: white powder, hygroscopic
- Density: 2.429 g/cm3
- Melting point: 235 °C (455 °F; 508 K)
- Boiling point: 452 °C (846 °F; 725 K)
- Solubility in water: 60.6 g/100 mL (0 °C); 145 g/100 mL (100 °C)
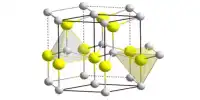
- Crystal structure: Isometric

Preparation
Ammonium bromide can be prepared by the direct action of hydrogen bromide on ammonia.
NH3 + HBr → NH4Br
It can also be prepared by the reaction of ammonia with iron(II) bromide or iron(III) bromide, which may be obtained by passing aqueous bromine solution over iron filings.
2 NH + FeBr2 + 2 H2O → 2 NH4Br + Fe(OH)2
Reactions
The chemical crystallizes into colorless prisms, possessing a saline taste. It sublimes on heating and is easily soluble in water. On exposure to air it gradually assumes a yellow color because of the oxidation of traces of bromide (Br–) to bromine (Br2).
Ammonium bromide is a weak acid with a pKa of ~5 in water. It is an acid salt because the ammonium ion hydrolyzes slightly in water.
Ammonium Bromide is strong electrolyte when put in water:
NH4Br(s) → NH4+(aq) + Br−(aq)
Ammonium bromide decomposes to ammonia and hydrogen bromide when heated at elevated temperatures:
NH4Br → NH3 + HBr
Uses
Ammonium bromide is used for photography in films, plates and papers; in fireproofing of wood; in lithography and process engraving; in corrosion inhibitors; and in pharmaceutical preparations.
- It is used in photography.
- It is used in fireproofing wood.
- It is used as a corrosive inhibitor