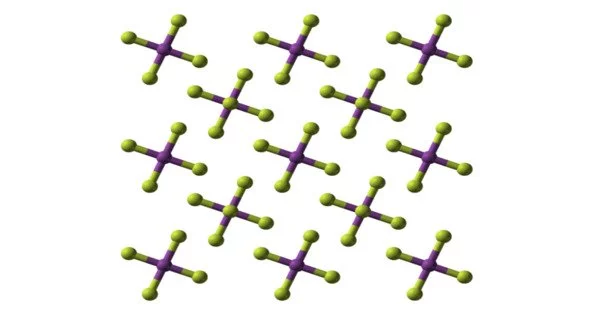
Bismuth pentafluoride is an inorganic compound with the formula BiF5. It is a pnictogen halide and a bismuth coordination entity. It is a white solid that is highly reactive. It derives from a hydride of a bismuthorane. The compound is of interest to researchers but not of particular value.
Properties
- Chemical formula: BiF5
- Molar mass: 303.97 g mol−1
- Appearance: long white needles, colorless crystalline solid
- Density: 5.40 g cm−3
- Melting point: 151.4 °C (304.5 °F; 424.5 K) , 154.4 °C
- Boiling point: 230 °C (446 °F; 503 K)
- Coordination geometry: octahedral Bi
Preparation
BiF5 can be prepared by treating BiF3 with F2 at 500 °C.
BiF3 + F2 → BiF5
In an alternative synthesis, ClF3 is the fluorinating agent at 350 °C.
BiF3 + ClF3 → BiF5 + ClF
Purification
Bismuth pentafluoride can be purified by sublimation under vacuum at around 100 ◦C in an apparatus made entirely of Vycor glass, which is not attacked by bismuth pentafluoride. It can also be crystallized from anhydrous HF prior to use.
Reactions
Bismuth pentafluoride is the most reactive of the pnictogen pentafluorides and is an extremely strong fluorinating agent. It reacts vigorously with water to form ozone and oxygen difluoride, and with iodine or sulfur at room temperature. BiF5 fluorinates paraffin oil (hydrocarbons) to fluorocarbons above 50 °C and oxidises UF4 to UF6 at 150 °C. At 180 °C, bismuth pentafluoride fluorinates Br2 to BrF3 and Cl2 to ClF.
BiF5 also reacts with alkali metal fluorides, MF, to form hexafluorobismuthates, M[BiF6], containing the hexafluorobismuthate anion, [BiF6]−.
When exposed to water, it produces highly toxic and corrosive hydrogen fluoride gas; it can ignite spontaneously; it is heated to decompose highly toxic fluoride smoke.
Storage, and Precautions
Bismuth pentafluoride is moisture sensitive. It should be stored in a tightly closed container and kept in a dry environment. It may cause irritation of the eyes, skin, digestive tract, and respiratory tract. The toxicological properties of bismuth pentafluoride have not been fully investigated.