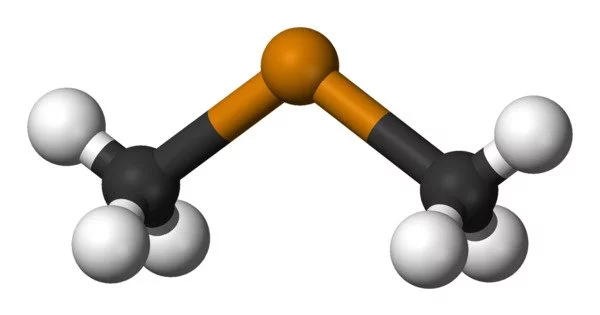
Dimethyl telluride, abbreviated DMTe, is an organotelluride compound with the formula (CH3)2Te. It is a colourless, volatile liquid with a garlic-like odour. This was the first material used in metalorganic vapour phase epitaxy to grow epitaxial cadmium telluride and mercury cadmium telluride. This compound belongs to the class of organotellurium compounds, which are distinguished by the presence of carbon-tellurium (C-Te) bonds.
Dimethyl telluride is best known for being produced by certain microorganisms, such as bacteria and fungi, during the tellurium metabolism process. Some plants also release it in trace amounts. The presence of this compound is frequently associated with tellurium-contaminated environments or as a byproduct of industrial processes.
Properties
One of the notable properties of dimethyl telluride is its distinctive smell, described as garlic-like or even akin to the smell of rotten cabbage. The smell is attributed to its tellurium content and is detectable even at very low concentrations.
- Chemical formula: C2H6Te
- Molar mass: 157.67 g·mol−1
- Appearance: Pale yellow, translucent liquid
- Odor: Garlic
- Melting point: −10 °C (14 °F; 263 K)
- Boiling point: 82 °C (180 °F; 355 K)
Discovery
Dimethyl telluride as a product of microbial metabolism was first discovered in 1939. It is produced by some fungi and bacteria (Penicillium brevicaule, P. chrysogenum, and P. notatum and the bacterium Pseudomonas fluorescens).
Toxicity
DMTe’s toxicity is unknown. When tellurium or one of its compounds is consumed, the body produces it. It is noticeable by the garlic-scented breath it gives those who are exposed, which is similar to the effect of DMSO. Tellurium is a known carcinogen.
Dimethyl telluride is not used commercially or industrially due to its toxicity and foul odour. In fact, it is considered hazardous and should be handled in a controlled environment with proper precautions. Dimethyl telluride, like all tellurium compounds, should be avoided because it can be harmful to human health.