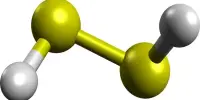
Potassium bifluoride is an inorganic compound with the formula K[HF2]. This colorless salt consists of the potassium cation (K+) and the bifluoride anion ([HF2]−). It is a chemical compound consisting of potassium, hydrogen, and fluorine. It is a white crystalline solid that is highly soluble in water.
The salt is used as an etchant for glass. Sodium bifluoride is related and is also of commercial use as an etchant as well as in cleaning products. The industrial production of fluorine entails the electrolysis of molten K[HF2] and K[H2F3]. The electrolysis of K[HF2] was first used by Henri Moissan in 1886.
Properties
- Physical Appearance: It is typically found as a white crystalline solid.
- Solubility: It is soluble in water, which means it can readily dissolve in aqueous solutions.
- Acidity: It is acidic in nature due to the presence of the bifluoride ion, which can release hydrogen ions in solution.
- Melting Point: It has a relatively high melting point, typically around 410°C (770°F).
- Hygroscopicity: It has a tendency to absorb moisture from the air, so it should be stored in a dry environment.
- Reactivity: It is reactive with many metals and metal oxides, forming fluorides upon reaction.
Synthesis and reactions
The salt was prepared by Edmond Frémy by treating potassium carbonate or potassium hydroxide with hydrofluoric acid:
2 HF + KOH → K[HF2] + H2O
With one more equivalent of HF, K[H2F3] is produced:
HF + K[HF2] → K[H2F3]
Thermal decomposition of K[HF2] gives hydrogen fluoride:
K[HF2] → HF + KF
Applications
- Metal surface treatment: It is used as an etchant for aluminum and its alloys. It removes oxides and other contaminants from metal surfaces prior to treatment or coating processes.
- Glass etching: It is utilized in glass etching processes due to its ability to dissolve silica. This property makes it useful in the production of decorative glassware and etched glass designs.
- Chemical synthesis: It is used as a source of fluoride ions in various chemical reactions, especially in organic synthesis where it can be employed as a mild fluorinating agent.
- Electronics manufacturing: It finds application in the electronics industry for cleaning and etching semiconductor materials and circuit boards.