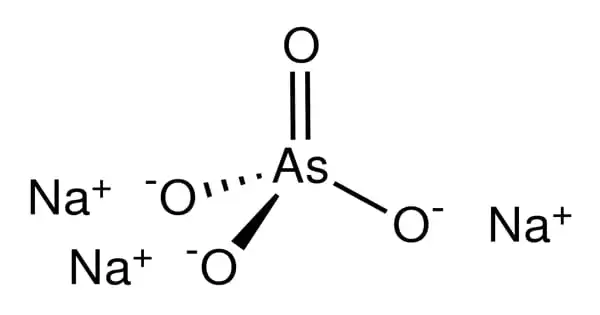
The inorganic chemical sodium arsenate has the formula Na3AsO4. It is a sodium inorganic salt. Related compounds, such as Na2HAsO4 (disodium hydrogen arsenate) and NaH2AsO4, are also known as sodium arsenate (sodium dihydrogen arsenate). It functions as an insecticide, antibacterial agent, herbicide, rodenticide, carcinogenic agent, antineoplastic agent, and antifungal agent. The trisodium salt is an extremely poisonous white or colorless substance. It is commonly referred to as the dodecahydrate Na3AsO4.12H2O. It is a molecular entity of arsenic and an inorganic sodium salt.
Sodium arsenite, aqueous solution appears as a solid’s aqueous solution. Toxic when ingested, inhaled, or absorbed via the skin. It is used as an antiseptic, in pesticides and herbicides, to preserve hides, and in the manufacture of colors.

Properties
Sodium arsenate is a chemical compound made up of sodium and arsenic. It is derived from arsenic acid. Arsenic is a chemical element with the symbol As and the atomic number 33. It is a deadly metalloid with numerous allotropic forms, including yellow (molecular non-metallic) and multiple black and grey forms (metalloids). Arsenic is found in three metalloidal forms with various crystal shapes in nature (the minerals arsenopyrite and the considerably rarer arsenolamprite and pararsenolamprite), although it is more usually found as a combination with other elements.
- Molecular Weight: 185.90668
- Appearance: White powder
- Melting Point: N/A
- Boiling Point: N/A
- Density: 2.8128 g/cm3
- Solubility in H2O: N/A
- Molar mass: 207.88851 g/mol
The compound can be obtained by neutralizing arsenic acid:
H3AsO4 + 3 NaOH → Na3AsO4 + 3 H2O
The salt (as its dodecahydrate) is isomorphous with trisodium phosphate. The anion AsO43- exists at high pH, but below pH 11.5, it converts to HAsO42- (also written HOAsO32-).
Sodium Metaarsenite is a trivalent arsenic-containing telomerase inhibitor with anticancer action that is extremely soluble and orally accessible. Although the precise mechanism by which sodium metaarsenite exerts its action is unknown, this drug appears to target and bind to telomeric sequences, specifically TTAGGG repeats, resulting in telomere shortening and subsequent induction of apoptosis and prevention of tumor cell proliferation.