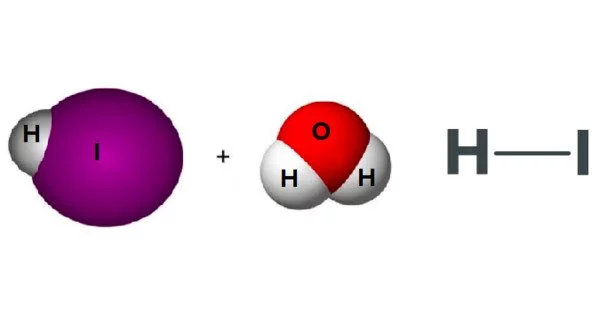
Aqueous hydrogen iodide (HI) solution is hydroiodic acid (or hydriodic acid). It is a colourless liquid that, when exposed to air, emits white fumes as a result of its reaction with atmospheric moisture. It is a strong acid, which means that it is completely ionised in an aqueous solution. It is extremely corrosive, causing severe burns and irritation to the skin, eyes, and respiratory system. It has no colour. Concentrated solutions typically contain 48% to 57% HI.
Properties
The odour of hydroiodic acid is strong and irritating. It is highly soluble in water and produces a strongly acidic solution. HI is a highly reactive compound that readily reacts with a variety of substances such as metals, organic compounds, and oxidising agents. Many materials, including metals, skin, and tissues, are corroded by it. It should be handled with caution, and when working with it, protective equipment should be worn.
- Chemical formula: HI(aq)
- Molar mass: 127.91 g/mol
- Appearance: colorless liquid
- Odor: acrid
- Density: 1.70 g/mL, azeotrope (57% HI by weight)
- Boiling point: 127 °C (261 °F; 400 K) 1.03 bar, azeotrope
- Solubility in water: Aqueous solution
Reactions
Hydroiodic acid is a powerful reducing agent and can react with various compounds. It is commonly used in organic chemistry as a source of iodide ions, which are important in many reactions. HI is often employed in the synthesis of organic compounds, such as pharmaceuticals and dyes.
Hydroiodic acid reacts with oxygen in air to give iodine:
4 HI + O2 → 2 H2O + 2 I2
Like other hydrogen halides, hydroiodic acid adds to alkenes to give alkyl iodides. It can also be used as a reducing agent, for example in the reduction of aromatic nitro compounds to anilines.
Uses
Hydroiodic acid has several applications, including as a reagent in chemical synthesis, as a reducing agent, and in the production of various iodine compounds.