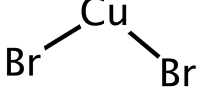
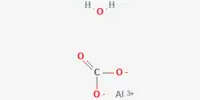
Aluminium(II) oxide, also known as aluminum monoxide, is a chemical compound composed of aluminum and oxygen with the formula AlO. It has been detected in the gas phase after aluminized grenade explosions in the upper atmosphere, as well as in stellar absorption spectra.
It is one of the aluminium oxides (the most common being Aluminium oxide Al2O3), and it is a rare example of an aluminium(II) compound because aluminium is normally found in its +3 oxidation state. However, this compound is not stable and readily reacts with oxygen to form aluminium oxide (Al2O3). Overall, aluminum(II) oxide is a versatile compound with a wide range of applications in various industries, including ceramics, electronics, and metallurgy.
Properties
It is a white or colorless solid that has a high melting point of 2,072 °C and is insoluble in water. In its natural form, it is found in the mineral corundum, which is a crystalline form of aluminium oxide. Due to its high melting point and chemical stability, aluminum oxide is commonly used as a refractory material in high-temperature applications such as furnace linings, crucibles, and refractory bricks.
- Chemical formula: AlO
- Molar mass: 42.98 g/mol
- Appearance: white or grayish-white powder.
- Density: 3.95 g/cm3
- Melting point: 2,072 °C
- Boiling point: 2,977 °C
- Solubility: insoluble in water and most organic solvents.
Application
Aluminium oxide is a commonly used compound in various industrial applications such as abrasives, refractories, ceramics, catalysts, and as a component in the production of aluminum metal. It is also used in the manufacturing of electrical insulators, high-temperature crucibles, and wear-resistant coatings.
It is often used as a support material for catalysts due to its high surface area and chemical stability. It is used as an abrasive in sandpaper and grinding wheels due to its hardness and durability. It is used in dental and medical applications such as dental implants and bone grafts due to its biocompatibility and strength.