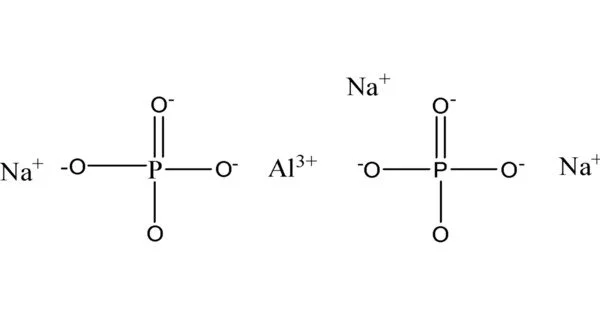
The inorganic compound sodium aluminium phosphate (SAlP) refers to sodium salts of aluminium phosphates. The formulas for the most common SAlP are NaH14Al3(PO4)8•4H2O and Na3H15Al2(PO4)8. Alumina, phosphoric acid, and sodium hydroxide are used to make these materials. It is an inorganic compound that is commonly used as a food additive. It is also known as sodium aluminum phosphate or sodium aluminum orthophosphate.
Sodium aluminum phosphate is generally considered safe for human consumption when used as a food additive within recommended limits. However, excessive consumption can lead to an accumulation of aluminum in the body, which has been associated with certain health risks.
Properties
- Chemical formula: NaH14Al3(PO4)8•4H2O
- Molar mass: 144.943 g/mol
- Appearance: white powder
- Odor: odorless
- Solubility in water: insoluble
- Solubility: soluble in HCl
- pH: In aqueous solutions, it has a pH of around 8-9.
- Density: The density is about 2.5 g/cm3.
- Melting point: It has a melting point of about 1000°C.
- Stability: It is stable under normal conditions, but can decompose at high temperatures.
In addition to the usual hydrate, an anhydrous SAlP, Na3H15Al2(PO4)8 (CAS#10279-59-1), is also known, with the ratio of phosphate to aluminum to sodium being 8:2:3. In addition, a SAlP with an unknown stoichiometry (NaxAly(PO4)z (CAS# 7785-88-8) is used.
Acidic sodium aluminum phosphates are used in baking powders as acids for chemical leavening of baked goods. SAlP reacts with baking soda to produce carbon dioxide when heated. The majority of its action occurs when the dough or batter is baked, rather than when it is mixed at room temperature. SAlPs are useful because they have a neutral flavor.
Application
Sodium aluminum phosphate is commonly used as a food additive (E541) in baking powders and as a leavening agent in bakery products. It is also used in the manufacturing of ceramics and glass, as well as in water treatment processes.
It is typically used as a leavening agent in baking powders, where it reacts with an acid (such as cream of tartar) to produce carbon dioxide gas, which causes baked goods to rise. It is also used as a stabilizer in some foods, such as cheese and pudding, to prevent them from breaking down or separating.
Safety
Sodium aluminium phosphate is generally recognized as safe by the U.S. Food and Drug Administration (FDA) when used in accordance with good manufacturing practices. However, excessive intake of aluminum has been associated with certain health risks, so it is important to consume it in moderation.